BCF Life Sciences,
European specialist in the extraction of free amino acids
We only sell what we produce.
We have been a pioneer in the circular economy since 1986.
Our products are aimed at the human, animal and plant health markets.
We are the only European specialist in the extraction and valorization of keratin from poultry feathers through biotechnologies.
Creation of
more than 100 direct jobs in 12 years
BCF Life Sciences has created nearly 100 direct jobs in 12 years, or a total of 550 jobs; in fact, one job created in industry generates 1.5 indirect jobs (by the activity of the factory) and 3 induced jobs (by the consumption of the employees hired).
A mastered know-how and
A manufacturing process
unique in Europe
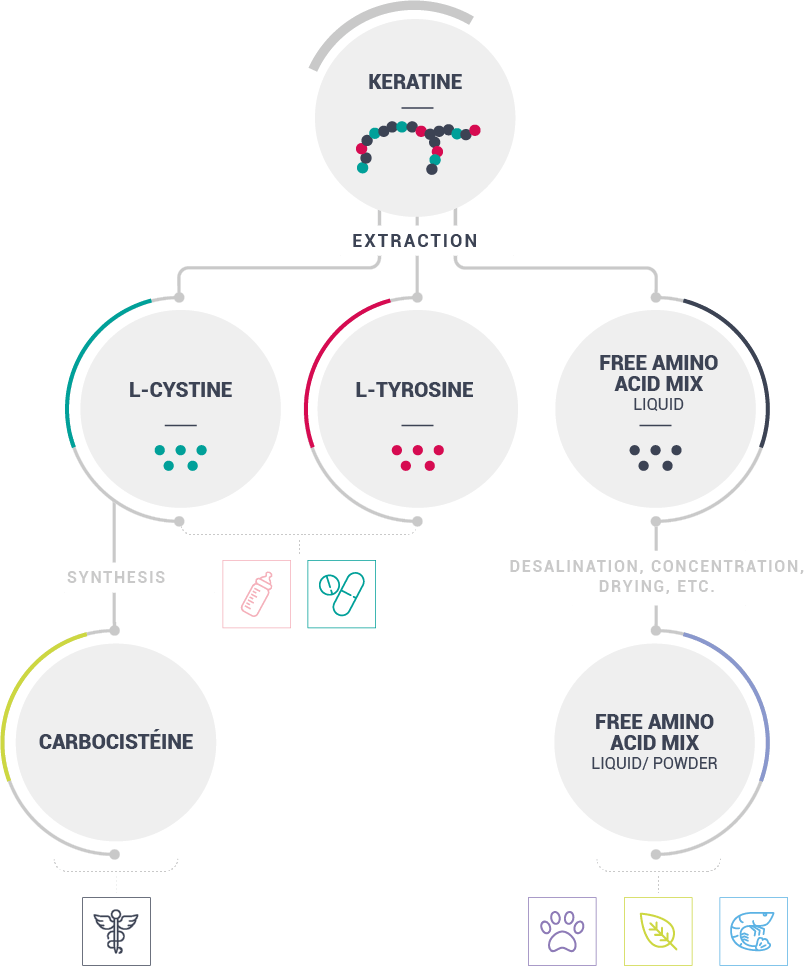
Created in 1986, BCF Life Sciences is the French specialist in the extraction of free amino acids3 from keratin, an animal protein naturally present in poultry feathers.
Our manufacturing process involves extracting and purifying two free amino acids,
L-Cystine4 and L-Tyrosine5, among the 17 present in the protein.
We also produce carbocistein6 and mixes of free amino acids.